🌡 Third law of thermodynamics
It’s about absolute zero temperature. Absolute zero (about -273 C) is the lowest possible temperature.
The third law of thermodynamics says:
It is not possible to reach a temperature of absolute zero.
Since temperature is a measure of molecular movement, there can be no temperature lower than absolute zero.
The third law of thermodynamics is related to the absence or lack of heat. How so?
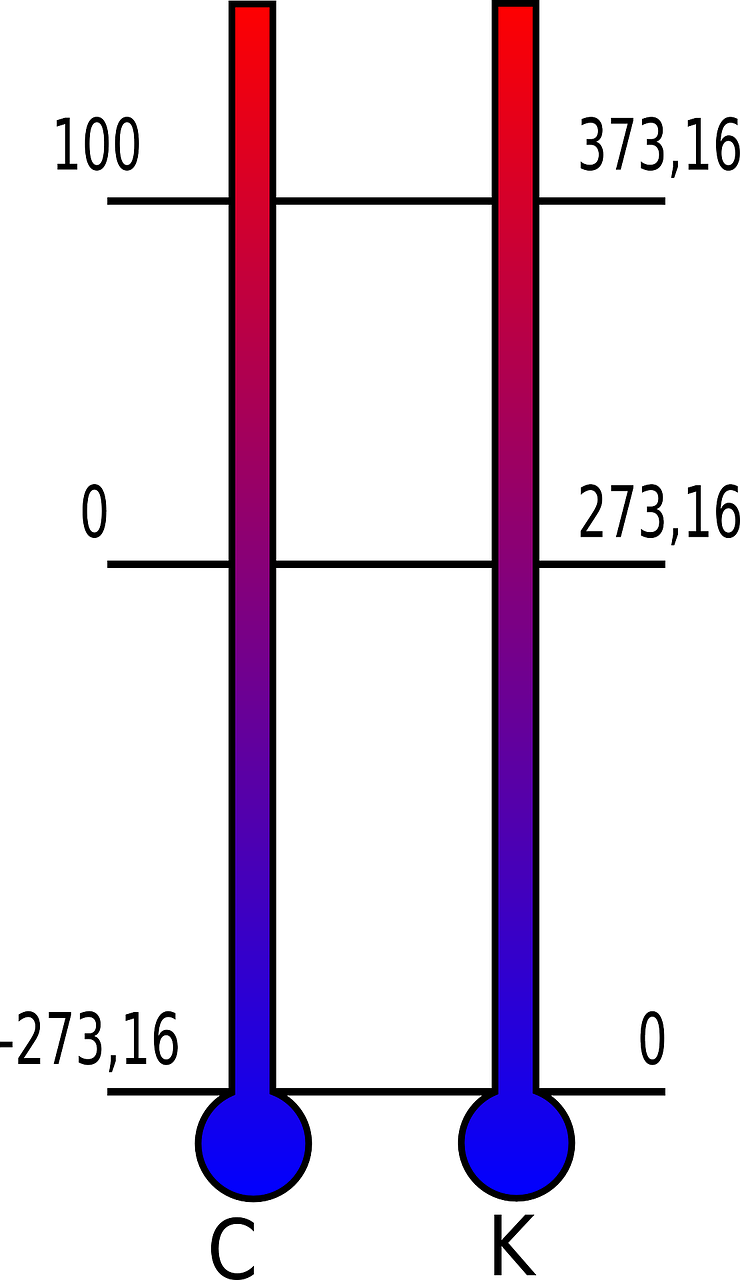 What is the temperature of a body and how is it measured? In fact, temperature of a body/object is a measurement of the process of movement of it molecules. When an object cools down (its temperature drops), its molecules begin to move more slowly. But there is a limit to which the temperature of a body can be lowered, and it cannot be colder than that. This is the absolute zero of temperature, which is the 0 Kelvin (0 K = -273.15 C = -459.67 F).
What is the temperature of a body and how is it measured? In fact, temperature of a body/object is a measurement of the process of movement of it molecules. When an object cools down (its temperature drops), its molecules begin to move more slowly. But there is a limit to which the temperature of a body can be lowered, and it cannot be colder than that. This is the absolute zero of temperature, which is the 0 Kelvin (0 K = -273.15 C = -459.67 F).
In other words, if a body manages to cool down to reach this temperature (0K), it will have zero energy.
In fact when the object is cooled down to the absolute zero temperature, it does not mean that the molecules in it have completely stopped moving and there is no movement at the molecular level.
Albert Einstein proved that even at a temperature of absolute zero in the cooled body there is still some energy.
Einstein also proved that the entropy of a body reaches its minimum when the body cools to absolute zero temperature. When a body begins to cool, its entropy begins to decrease, and at a temperature equal to 0K its entropy is minimal. This also means that its degree of decay and disorder also decreases as it cools.

The third law of thermodynamics explains what happens to bodies when they cool so much that their temperature reaches 0 K.
🔍 👀 Observe this – frozen crystal >>>
Good to see also:
First law of thermodynamics
Second law of thermodynamics
Zeroth law of thermodynamics


