First law of thermodynamics
First law of thermodynamics is about the conservation of energy.
The first law of thermodynamics says:
Energy can be changed from one form to another, but it cannot be created or destroyed. The total amount of energy and matter in the Universe remains constant.
The law states that the energy input for a system equals the total energy output, which is the sum of the useful work and the waste heat energy.
The change in internal energy (U) of a system is equal to the heat added to the system (Q), minus the work done by the system (W):
U=Q-W
Examples: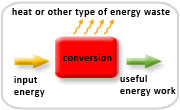
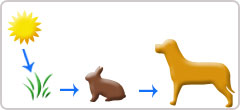
Shown are two examples of energy being transferred from one system to another and transformed from one form to another. Plants can convert electromagnetic radiation (light energy from the sun) into chemical energy (photosynthesis). Animals can convert the chemical energy in food into kinetic energy thаt allows them to move and run.
See also:
Third law of thermodynamics
Second law of thermodynamics
Zeroth law of thermodynamics


